Chemical Bonding MCQs – Ionic, Covalent & Metallic Bonds
Practice 10 MCQs on chemical bonding covering ionic bonds, covalent bonds, metallic bonds, Lewis structures, polarity, and bond properties.
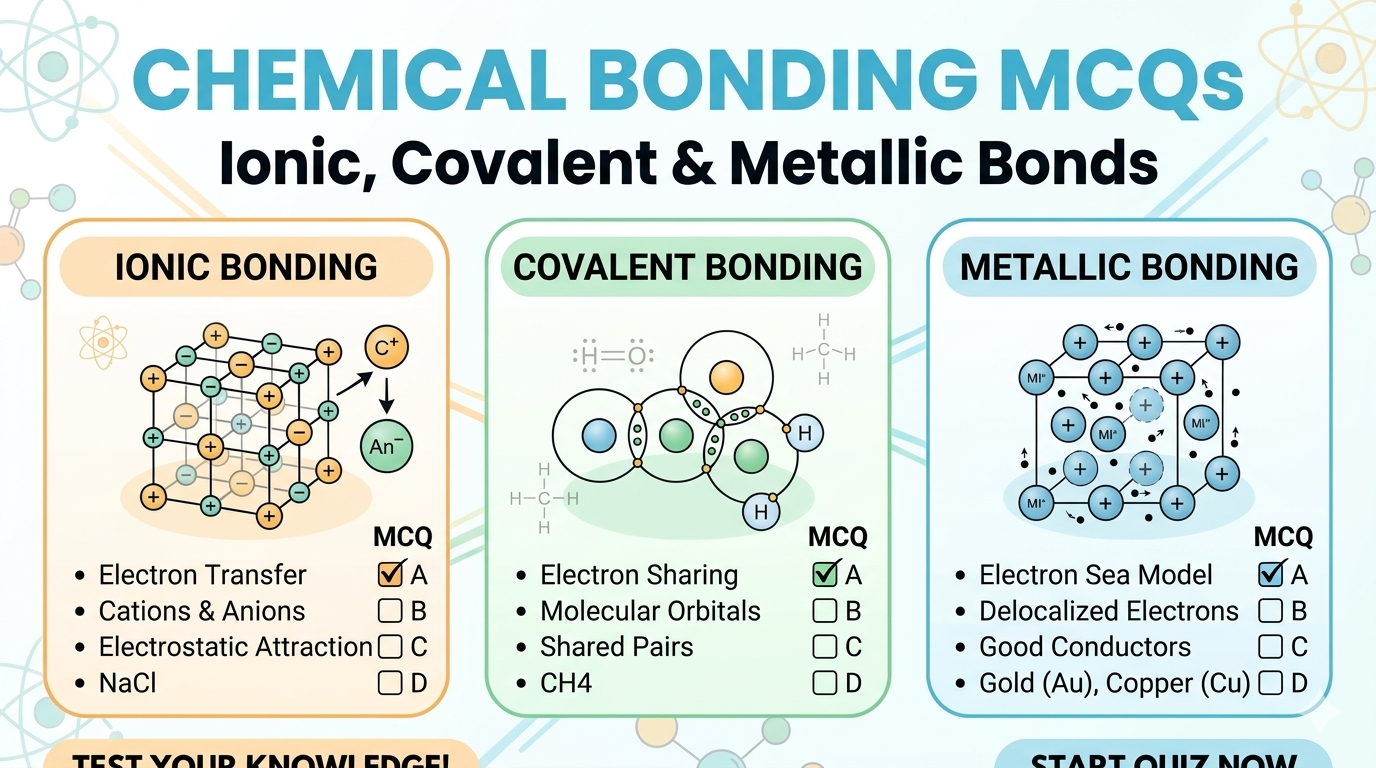
Ionic bonds form when one atom transfers electrons to another, creating oppositely charged ions (cation + anion) that attract each other. Example: NaCl — Na gives 1e⁻ to Cl.
Covalent bonds form when two non-metal atoms share electrons. Both atoms achieve a stable electron configuration. Examples: H₂, H₂O, CO₂, CH₄. Ionic bonds form between metals and non-metals.
NaCl is an ionic compound. Sodium (metal) loses 1 electron to become Na⁺; Chlorine (non-metal) gains it to become Cl⁻. The electrostatic attraction between Na⁺ and Cl⁻ is the ionic bond.
A double bond consists of 2 shared electron pairs (4 electrons total). Single bond = 1 pair (2e⁻), double = 2 pairs (4e⁻), triple = 3 pairs (6e⁻). Example: O₂ has a double bond (O=O).
N₂ (nitrogen gas) has a triple bond (N≡N) — 3 shared electron pairs. This makes N₂ very stable and unreactive. Triple bonds are shorter and stronger than single or double bonds.
If electronegativity difference > 1.7, the bond is considered ionic (electron transfer). If 0.4–1.7, it’s polar covalent. If < 0.4, it's nonpolar covalent. This is Pauling's guideline.
H₂O has a bent/V-shape due to 2 bonding pairs and 2 lone pairs on oxygen. Lone pairs repel more than bonding pairs, pushing the H-O-H angle from 109.5° to 104.5°. This is explained by VSEPR theory.
Metallic bonds exist between metal atoms in a lattice. Valence electrons are delocalized — they form an ‘electron sea’ surrounding positive metal ions. This explains conductivity, malleability, and luster.
Triple bonds have the highest bond energy (strongest) and shortest bond length. Bond energy order: Triple > Double > Single. More electron pairs mean stronger attraction and more energy needed to break the bond.
Polar covalent bonds form when electrons are unequally shared due to a difference in electronegativity between atoms. The more electronegative atom gets a partial negative charge (δ⁻). Example: H-Cl, H-O.
About These Chemical Bonding MCQs
These Chemical Bonding MCQs cover ionic, covalent, and metallic bonds with questions on electronegativity, bond polarity, molecular shapes, and Lewis structures.
Chemical bonding is a core FSc and MDCAT Chemistry topic. Understanding why and how atoms bond is fundamental to all of chemistry.
Topics Covered
- Ionic bonding — electron transfer
- Covalent bonding — electron sharing
- Single, double, and triple bonds
- Polar vs nonpolar covalent bonds
- Molecular geometry (VSEPR theory)
- Metallic bonding and electron sea model
MDCAT Preparation Focus
Know the electronegativity difference thresholds (ionic > 1.7, polar covalent 0.4–1.7, nonpolar < 0.4). Practice drawing Lewis dot structures and applying VSEPR theory to predict molecular shapes.